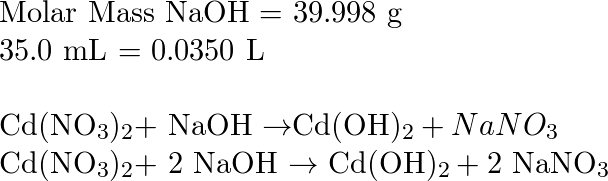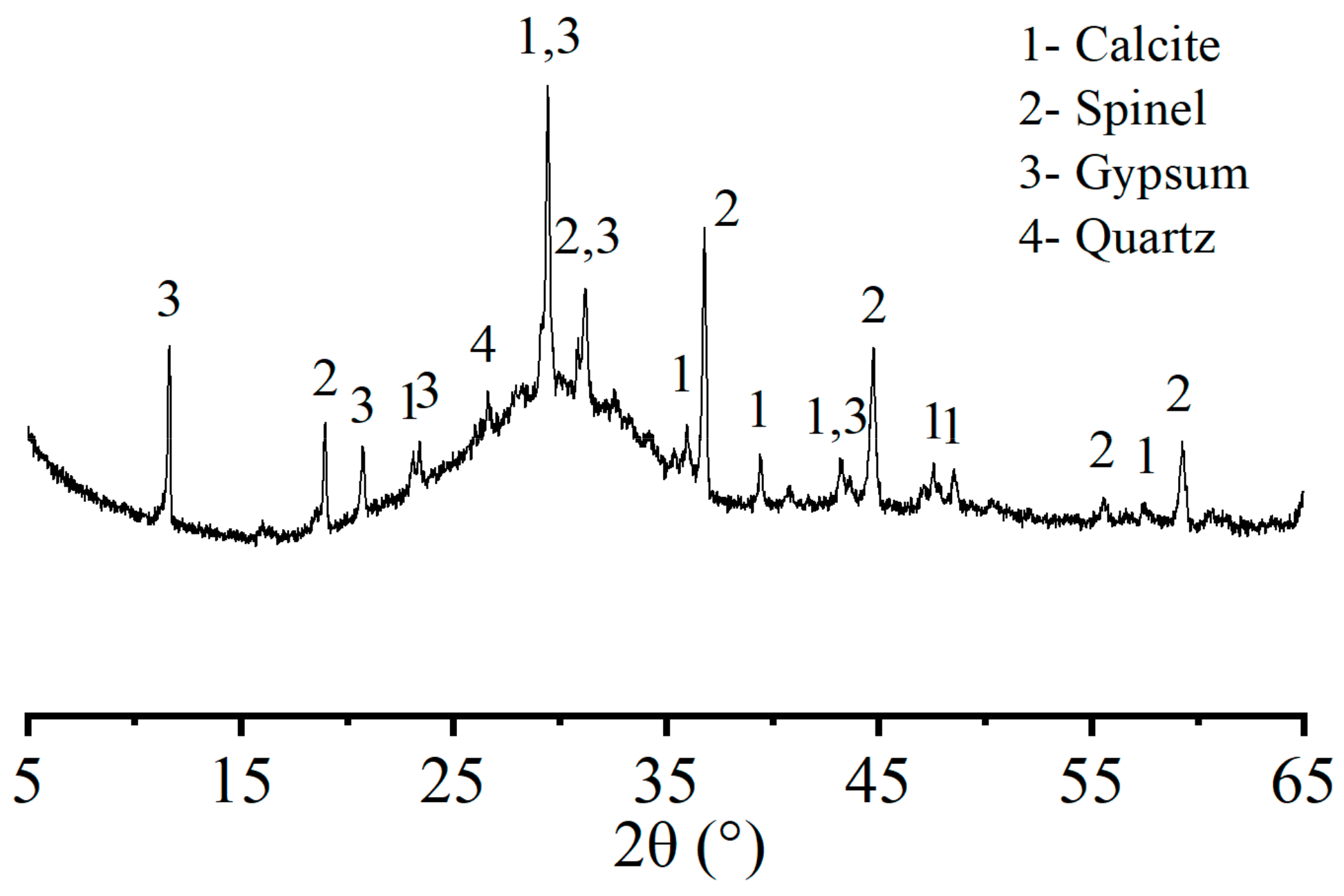
Crystals | Free Full-Text | Effect of Different NaOH Solution Concentrations on Mechanical Properties and Microstructure of Alkali-Activated Blast Furnace Ferronickel Slag
1, 2*Rachtanapun, P., 1Simasatitkul, P., 1Chaiwan, W. and 1Watthanaworasakun, Y. Effect of sodium hydroxide concentration on pro

Given the cell: Cd(s)|Cd(OH)2(s)|NaOH(aq, 0.01 M)|H2(g, 1 bar)|Pt(s) with Ecell = 0.0 V . If E^oCd^2 + |Cd = - 0.39 V , then Ksp of Cd(OH)2 is:

PDF) Influence of NaOH on structural, morphological and band gap analysis of CdS nanoparticles | Sakthivishnu Rangaswamy - Academia.edu

a) CD spectra for ligand 4 in basic media (2 equiv. of NaOH). (b) CD... | Download Scientific Diagram

Insights into the removal of Cd and Pb from aqueous solutions by NaOH–EtOH-modified biochar - ScienceDirect

SOLVED: A 0.35 g sample of NaOH is dissolved in water and added to 1 L of wastewater solution containing 0.454 g of CdClz, in an attempt to remove the toxic cadmium(II)

Given the cell: Cd(s)|Cd(OH)2(s)|NaOH(aq, 0.01 M)|H2(g, 1 bar)|Pt(s) with Ecell = 0.0 V . If E^oCd^2 + |Cd = - 0.39 V , then Ksp of Cd(OH)2 is:

OneClass: What mass of NaOH is needed to precipitate the Cd2+ ions from 35.0 mL of 0.460 M Cd(NO3)2 s...

Given the cell: Cd(s)|Cd(OH)_2(s)|NaOH(aq,0.01M)|H_2(g,1bar)|Pt(s) with E_(cell)=0.0V.ifE_(Cd^(2... - YouTube

OneClass: 1. Let us assume that Cd(OH)2(s) is completely insoluble, which signifies that the precipit...

![ANSWERED] If 4 moles of NaOH react with 1 mole of C... - Physical Chemistry ANSWERED] If 4 moles of NaOH react with 1 mole of C... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/47412038-1658679986.4827924.jpeg)