Single and mixture toxicity of As, Cd, Cr, Cu, Fe, Hg, Ni, Pb, and Zn to the rotifer Proales similis under different salinities - ScienceDirect
The coordination chemistry of Zn(ii), Cd(ii) and Hg(ii) complexes with 1,2,4-triazole derivatives - Dalton Transactions (RSC Publishing)
![Energy analysis of metal–metal bonding in [RM–MR] (M = Zn, Cd, Hg; R = CH3, SiH3, GeH3, C5H5, C5Me5) - ScienceDirect Energy analysis of metal–metal bonding in [RM–MR] (M = Zn, Cd, Hg; R = CH3, SiH3, GeH3, C5H5, C5Me5) - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022328X06009089-fx1.jpg)
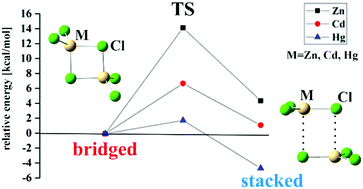
Energy analysis of metal–metal bonding in [RM–MR] (M = Zn, Cd, Hg; R = CH3, SiH3, GeH3, C5H5, C5Me5) - ScienceDirect

Anion⋅⋅⋅Anion Attraction in Complexes of MCl3− (M=Zn, Cd, Hg) with CN− - Wysokiński - 2020 - ChemPhysChem - Wiley Online Library

Numerical overview of CSD data. Sp = Zn, Cd or Hg, X = any atom, all... | Download Scientific Diagram

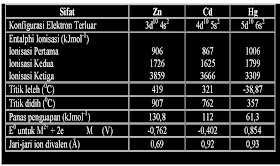
a). Why IE1 of Zn, Cd and Hg are very high? (b). The relative difference in IE of any two consecutive d-bloch elements is smaller than those of s and p-block elements. (

why are Zn,Cd & Hg not considered as transition elements | part 1 | Unit-8 |cbse|class 12 chemistry - YouTube
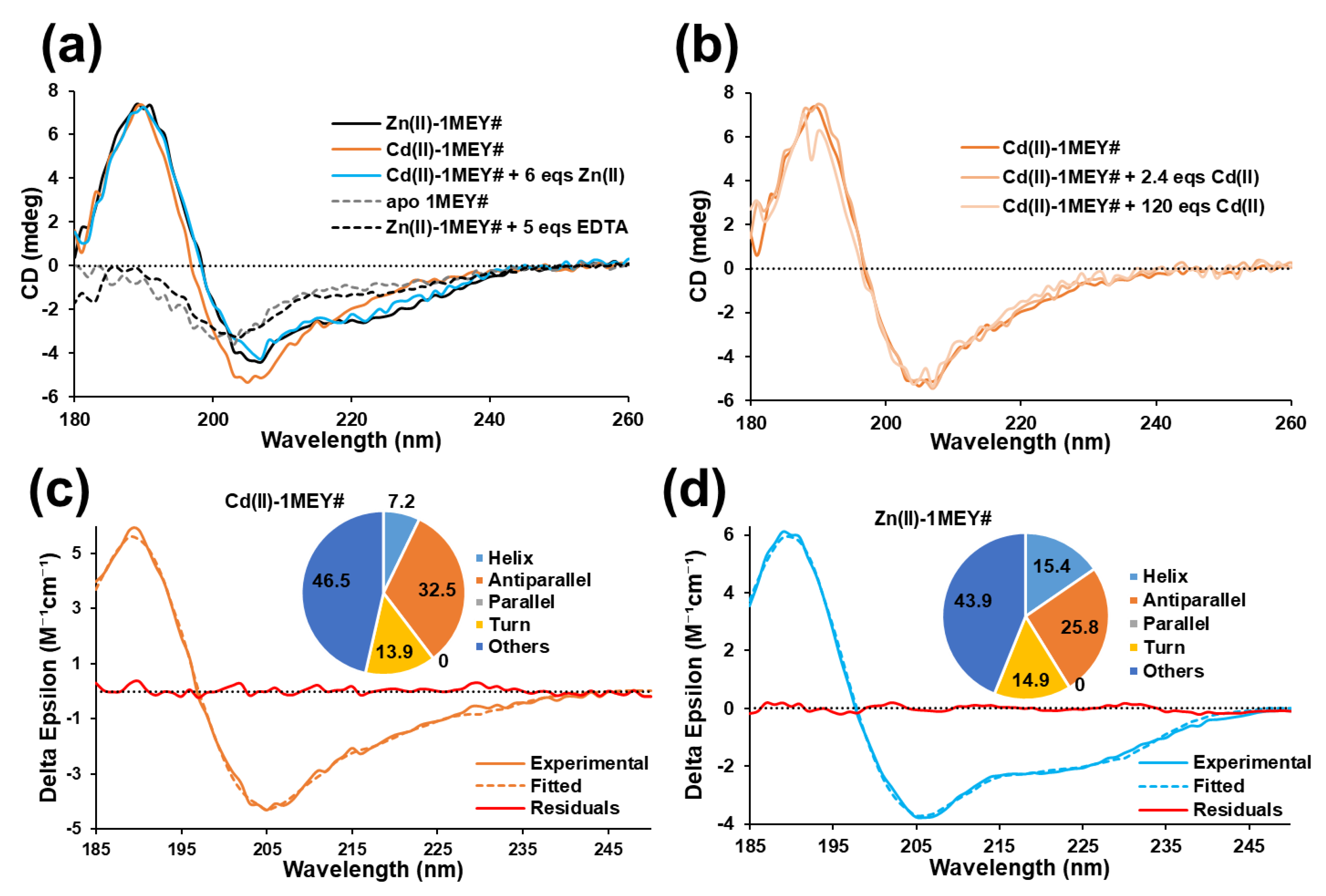
Inorganics | Free Full-Text | Interactions of an Artificial Zinc Finger Protein with Cd(II) and Hg(II): Competition and Metal and DNA Binding
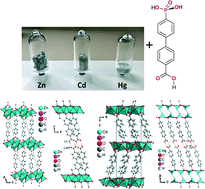
Syntheses, structures and properties of group 12 element (Zn, Cd, Hg) coordination polymers with a mixed-functional phosphonate-biphenyl-carboxylate linker - CrystEngComm (RSC Publishing)

Complex cubic metallides AM~6 (A=Ca, Sr; M=Zn, Cd, Hg). Synthesis, crystal chemistry and chemical bonding

The names transition metals and inner transition metals are often used to refer to the elements of d-and f-blocks respectively. Zn, Cd and Hg are usually. - ppt download